Let's try to clarify the types of tests for Sars-CoV-2
We often find ourselves having to deal with technical terminologies used in an improper or confused way. We will now try to clarify the meaning of the main terms using the material produced by the Istituto Superiore di Sanità.
Diagnostic devices are classified into different types depending on whether or not to use equipment to perform the test, the type of marker that is detected and the person who will have to use the test.
Classification based on the use or not of equipment
- QUICK TEST: only tests that do not require equipment to obtain the result are intended. It has nothing to do with tampon, salivary, lancing or anything else and neither does the method of execution have anything to do with it.
- NOT QUICK TESTS: we only mean tests that require equipment to obtain the result. A classic example is the molecular buffer, but not only.
Here we already understand that the term rapid is improperly used in relation to the speed of execution but this is not the case. Some tests may provide fast results and be designed in the form of portable equipment but do not fall within the European Commission's definition of "rapid tests". For example, commercial tests that adopt “NAT” molecular methods are considered not rapid even if they are able to ensure a faster result than conventional tests.
Assays that detect the presence of virus components (RNA or antigen)
These assays are typically performed on nasopharyngeal (nasopharyngeal) swab, saliva or bronchoalveolar lavage and there are two types:
- Molecular methods "NAT": detect the presence of viral RNA in the biological sample through the identification of specific genomic sequences characteristic of the virus and their cyclic amplification, making them detectable.
- Antigen research methods: immunoenzymatic methods that detect the presence of the virus in the sample by recognizing specific virus surface proteins, using specific antibodies.
Serological (or antibody) assays:
These assays are performed on whole blood, serum or plasma. They are indirect methods as they do not detect the presence of the virus, but detect the presence of antibodies produced in response to exposure to the virus.
Classification based on the intended user
- Tests for professional use: include both tests intended for use in a laboratory and rapid tests used by healthcare personnel.
- Self-diagnostic tests: these are devices intended to be used by the patient himself in the home
Biological classification for Covid-19 diagnosis
For the diagnosis of SARS-CoV-2 infection, the sample of choice is a respiratory tract sample. This can be of the upper respiratory tract (nasal swab, oral-pharyngeal swab, rhino-pharyngeal swab) and more rarely, where available, of the lower respiratory tract: endo-tracheal aspirate or broncho-alveolar lavage. The lower respiratory tract sample is preferred for a higher virus concentration in cases of pneumonia.
- Lower respiratory tract samples: Lower respiratory tract samples are collected in a sterile container, wearing the appropriate PPE and minimizing the possibility of generating aerosols. For this reason, induced sputum sampling is not recommended.
- Nasopharyngeal and oropharyngeal swabs: The collection of the nasopharyngeal and oropharyngeal swab is a procedure that consists in the removal of mucus that covers the superficial cells of the mucous membrane of the nasopharynx or oropharynx, using a swab (currently there are swabs in synthetic material capable of retaining much more organic material than cotton wool pads).
Procedures for performing swabs for Sars-CoV-2 diagnostics
- Nasopharyngeal swab (photo 1): the nasopharynx is located deep behind the nose and is the upper portion of the pharynx, which divides from top to bottom into the nasopharynx, oropharynx and hypopharynx. To be reached, it is necessary to advance the swab into the nasal cavities perpendicular to the patient's face, resting it on the floor of the nasal cavities, for an average length of 8 to 12 cm.
- Oropharyngeal swab (photo 2): the oropharynx is placed behind the oral cavity and includes the tonsillar region, the soft palate, the uvula, the base of the tongue and the lateral and posterior wall of the oropharynx, these latter being placed behind the regions tonsils and uvula. The swab of the oropharynx involves rubbing the tonsillar regions and the posterior wall of the pharynx gently, without touching the tongue, cheeks and dental arches.
- Anterior nasal swab (photo 3): it requires its introduction into the nasal cavities to reach the mucous membranes where the virus colonizes. In the anterior nasal swab material is taken at the level of the first 2 cm of the nose by gently touching the mucosa with the swab in a circular motion and leaving it in place for a few seconds to soak up the secretions.
- Nasal swab of the middle turbinate (photo 4): in the lateral wall of the nose, from bottom to top, three turbinates are described, inferior, middle and superior, which are structures covered by mucosa where the virus can colonize. The tampon, to reach the middle turbinate, must be oriented upwards and laterally inclined by about 45 ° for no more than 3 cm, with a delicate contact of these structures.
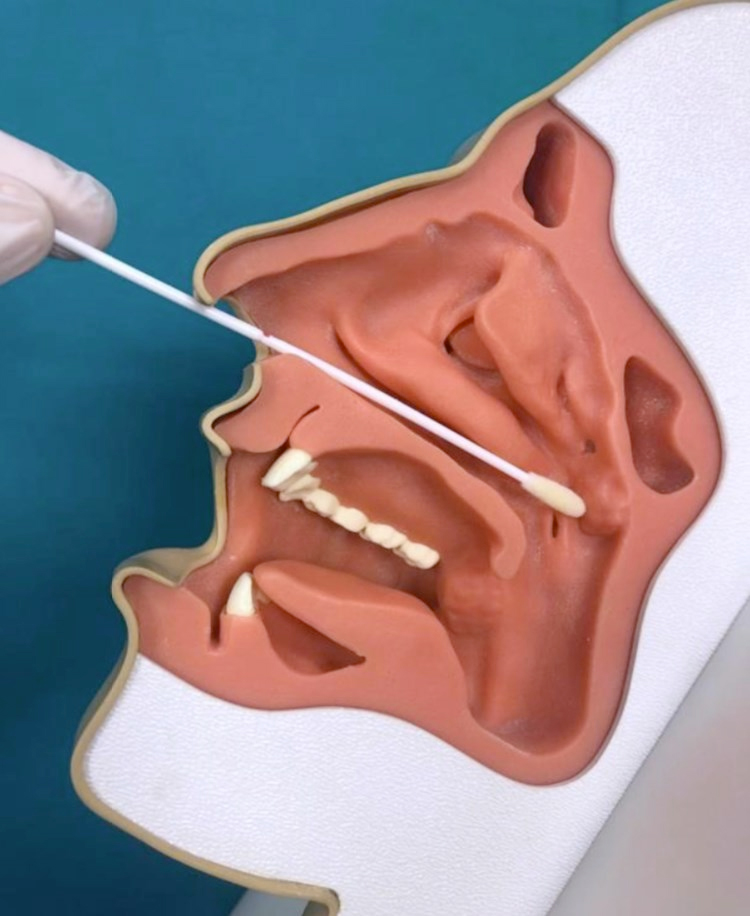
Photo 1
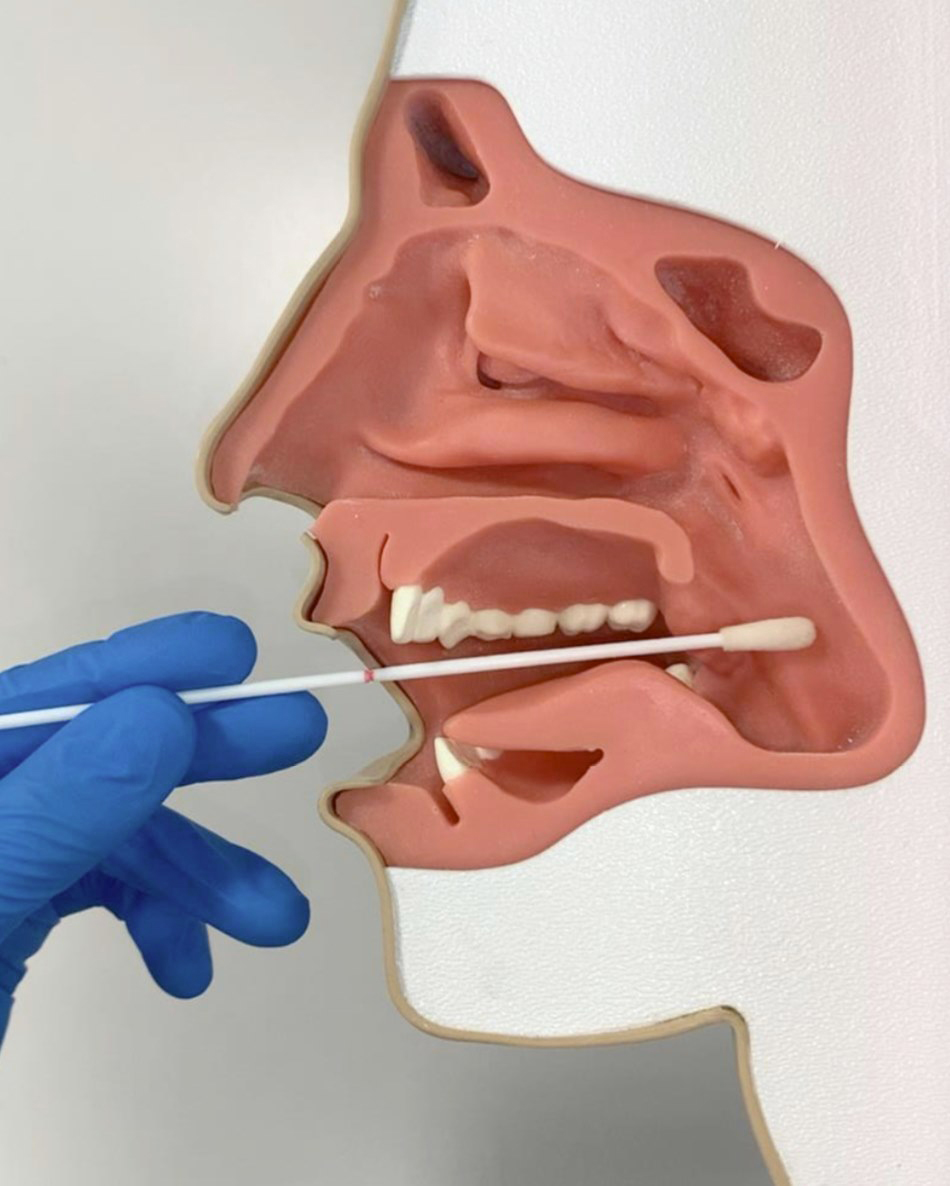
Photo 2
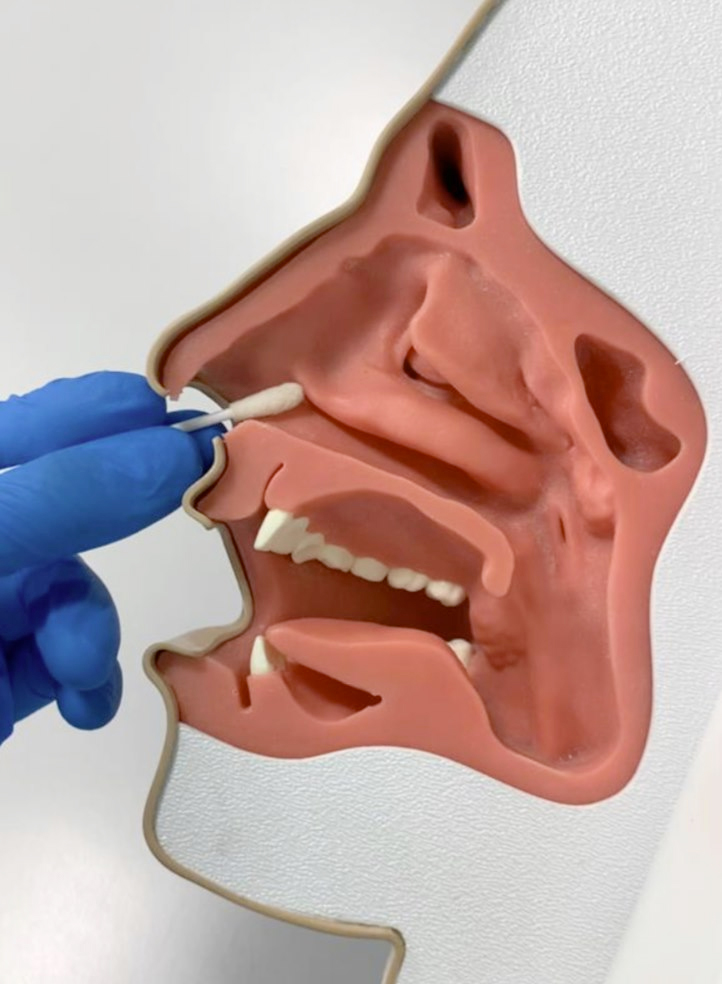
Photo 3
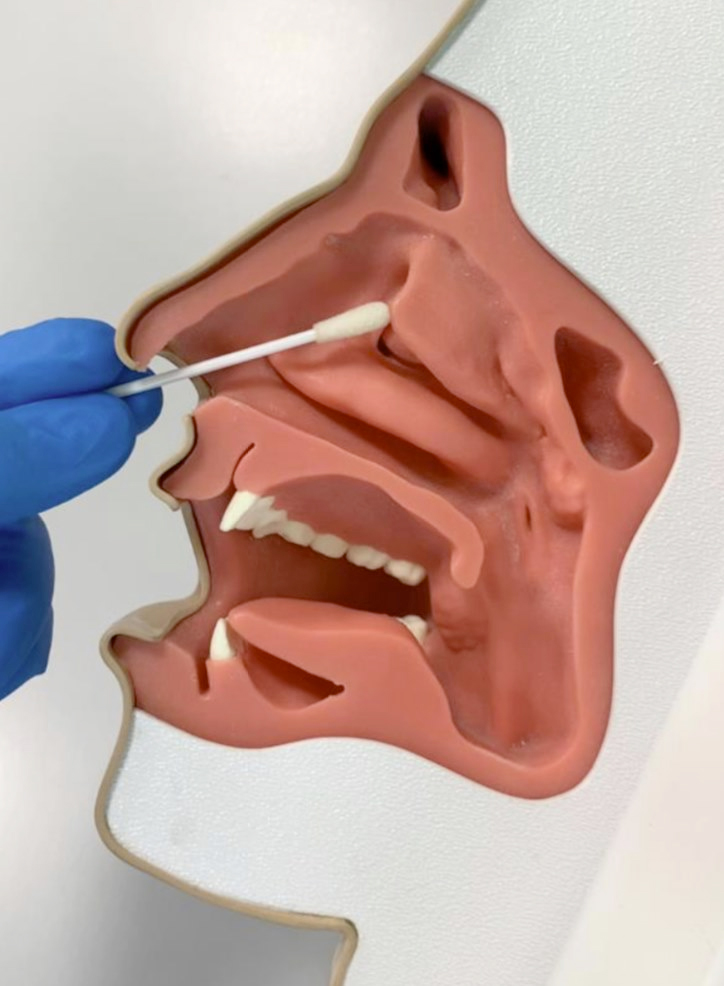
Photo 4
Sources:
1. ISS COVID-19 report n. 11/2020 Rev. 2 - Interim recommendations for the correct collection, storage and analysis on rhino / oropharyngeal swab for the diagnosis of COVID-19
2. ISS COVID-19 report n. 54/2020 - Technologies to support proximity detection: reflections for citizens, professionals and stakeholders in the COVID-19 era

