Comparison between the human genome contained in Priorix Tetra and in the MRC-5 cell line

Brief presentation of the results
We present today a further very important study on the genomic sequencing of the cell line found within the quadrivalent vaccine MPRV (measles, rubella, mumps and varicella) Priorix Tetra, and that of the MRC-5 cell line (which is the one declaredly used in the development of the vaccine itself).
Summarizing the previous published reports:
- we first demonstrated how the amount of DNA present in the aforementioned vaccine was well above the allowed;
- we then verified that the cell line is actually the declared MRC-5;
- we subsequently proceeded to genome sequencing of the cell line extracted directly from the vaccine, highlighting very heavy mutations that entail the possibility of serious health risks in the use of this vaccine.
What we present here is precisely the comparison work between the two genomes: what we really found in the vaccine and that of the MRC-5 purchased from the ATCC database.
The result is that the two lines present important differences from the point of view of genetic stability. In particular, the vaccine genome appears to be heavily modified compared to that of the cell line deposited in 1966.
This means that the producers, in this case of the Priorix tetra, have purchased the cell line in optimal conditions from the point of view of genetic stability and with continuous use over time for the purpose of production have put on the market vaccines containing human genetic material progressively increasingly modified and dangerous for the health of the vaccinated.
Since regulatory agencies do not require periodic control of the genetic stability of fetal lines and do not even set a minimum safety threshold, we can deduce that a fundamental requirement for the quality and safety of the vaccines that have been obtained from human cell lines, that is, the need to verify that they do not acquire potentially dangerous mutations and rearrangements and to guarantee the elimination of processing residues of genetic material if present.
It is not for us to identify which part of the production or conservation process can affect the quality and safety of the product, but what we can testify today by publishing this report is that it is urgent for control bodies to carry out a thorough examination of the results that our analyzes have highlighted. in these two years, focusing attention on the finished product, sold and administered and not only on the individual authorized "ingredients".
Remember once again that these products are part of the Italian vaccination calendar and are currently compulsorily administered to the pediatric population of our country as well as other European countries and in general all over the world.
The control bodies have an enormous responsibility for safety which is currently being heralded in words but which in fact, apparently, and as it appears from our analyzes, cannot be guaranteed and is indeed clearly disregarded.
Circular visualization of genomes (circos plot)
A graphical representation of the two genomes called 'circos plot' is shown below.
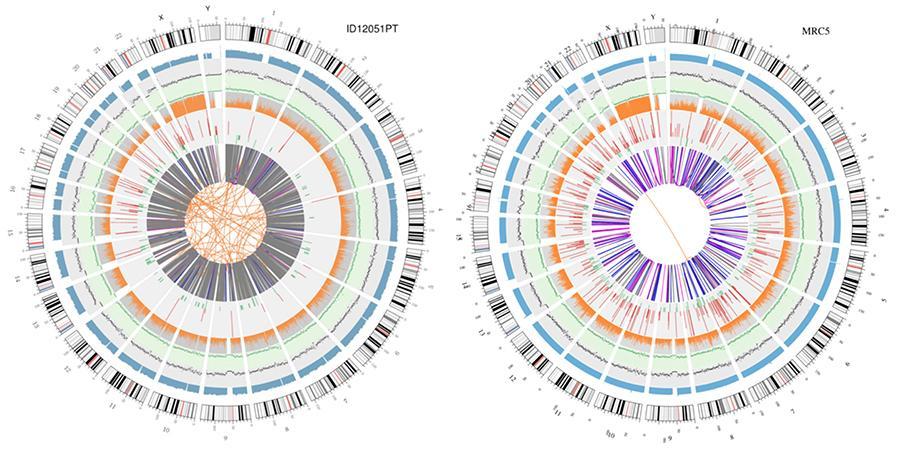
Meaning of the various concentric circles
- The outer circle (the first circle) is the chromosome number.
- The second ring (blue) represents the coverage of histogram-style reads. Each histogram is the average coverage of an area of 0,5 Mbp.
- The third ring (black) represents the density of the INDELs in a 'dispersion graph' style. Each black point is calculated as the number of INDELs (Small insertions / deletions) in a range of 1 Mbp.
- The fourth ring (green) represents the density snp in a 'dispersion graph' style. Each green dot is calculated as the number of SNPs (single nucleotide variants) in a range of 1 Mbp.
- The fifth ring represents the proportion of homozygosity (orange) and heterozygosity (gray) SNP in histogram style. Each histogram is calculated from a 1 Mbp region.
- The sixth ring represents the inference of CNV (Variants in number of copies). Red means gaining pieces of DNA and green means loss.
- The most central ring represents the inference of SV (Structural Variants) in the exonic and splicing regions. BETWEEN (orange, translocations), INS (green, insertions), DEL (deletions, gray), DUP (duplications, pink) and INV (inversions, blue).
Conclusions
The two genomes show important differences. In particular, the vaccine genome turns out to be heavily changed compared to that of the cell line deposited in 1966. It must be said that we did not have the possibility to sequence the cell line that is used by GlaxoSmithKline for the production of the Priorix Tetra vaccine, but that of the stock original. Pharmaceutical companies that use cell lines for the production of drugs and vaccines, have their MRC-5 line but this does not change our suspicions, that is the fact that the control of genetic stability is not required periodically and this is very serious in our opinion , above all because it is allowed to be present without a limit.
Download: CORVELVA-comparison-human-genome-Priorix-Tetra-and-line-MRC-5.pdf
Comparison between human genome contained in Priorix Tetra and in the MRC-5 cell line (ATCC standard MRC-5 ATCC® CCL-171 ™)
The presence of the rubella virus was demonstrated by sequencing an extremely deep RNA-seq library (approximately 260 million Illumina sequences produced). 114 out of 260 million sequences were detected, equal to 0.00004% of the total sequence. The rubella genome sequences were then confirmed manually with the BLAST software (Basic Local Alignment Search Tool, https://blast.ncbi.nlm.nih.gov/Blast.cgi). The sequenced library at a lower depth (about 12 million Illumina paired sequences, equal to 6 million sequenced library fragments) had not detected the presence of ANY reads attributable to rubella in this lot.
The sequences of the other viruses present in the vaccine (chickenpox, measles and mumps) were also validated in the same manual way, confirming that they were correctly assigned.
Introduction
The new generation sequencers have become instruments of choice for in-depth analyzes in the field of biology and medicine, especially the precision one. These tools allow a new and more global approach to a series of applications such as de novo sequencing, metagenomics, epigenomics, transcriptome sequencing and genome re-sequencing.
This latter application (re-sequencing) is very widespread in the human field both for research and diagnostic purposes and consists in the sequencing with NGS (Next Generation Sequencing) technology of an entire individual genome in order to map single nucleotide mutations (SNP, pronunciation 'snip '), insertions and deletions of more or less long sequences occurred in certain positions of the genome and variations in the number of copies of portions of genome / genes (CNV, Copy Number Variants).
The sequencing procedure requires that an individual's DNA is mechanically broken into small fragments (400-500 base pairs) and fragments of artificial DNA tracts called adapters are tied to the fragments, which allow to bind the fragments of human DNA to a glass surface on which the bases are then read (A, C, G, T). The DNA bases are read by incorporating chemical reactions of nucleotides labeled with fluorescent molecules. The millions of sequences (reads) obtained from sequencing on the glass surface are then mapped on the human reference genome with appropriate software and then all the variants present in the analyzed genome compared to the reference are identified.
This same procedure was performed on the human genome present in Priorix Tetra lot. n. A71CB256A and on the DNA extracted from the MRC-5 cell line, deposited in 1966 at ATCC (MRC-5 ATCC® CCL-171 ™).
ATCC is the world's leading organization of biological materials resources and standards whose mission focuses on the acquisition, authentication, production, storage, development and distribution of reference microorganisms, cell lines and other standard biological materials. It was established in 1925, when a committee of scientists recognized the need for a centralized collection of biological materials that could serve scientists around the world.
The MRC-5 cell line is derived from normal lung tissue from a 14-week-old male fetus and was deposited in ATCC by JP Jacobs in September 1966. These cells are said to be able to double 42 to 46 populations before initiation. of senescence. The reference article for this cell line dates back to 1970: https://www.nature.com/articles/227168a0
Results
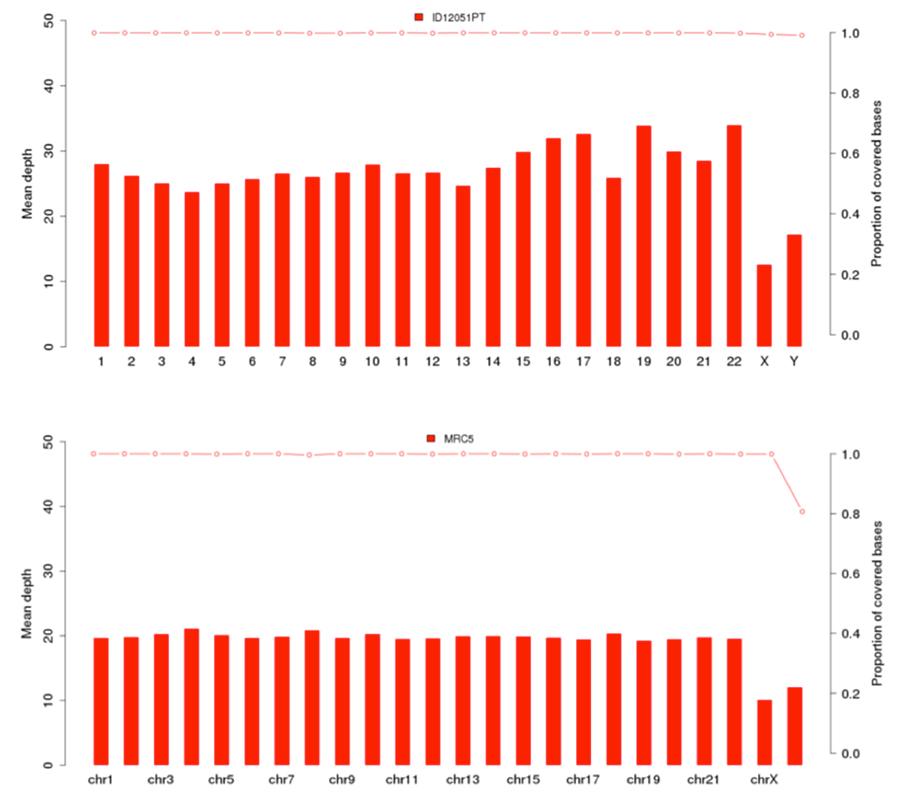
As previously shown, the human DNA present in Priorix Tetra lot. n. A71CB256A (indicated in the following graphs as sample ID12051PT) is a complete individual genome, that is, there is genomic DNA of all the chromosomes of a male individual. The coverage graphs (Mean depth = average coverage) of the 22 human chromosomes and the X and Y chromosomes are shown below, for the vaccine (image above) and the cell line MRC5 (image below).
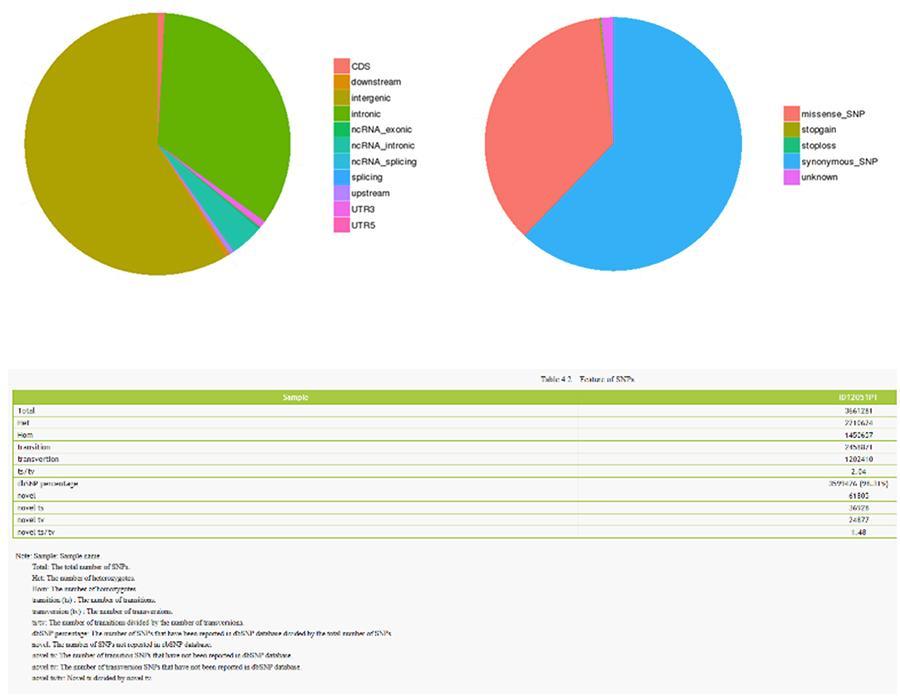
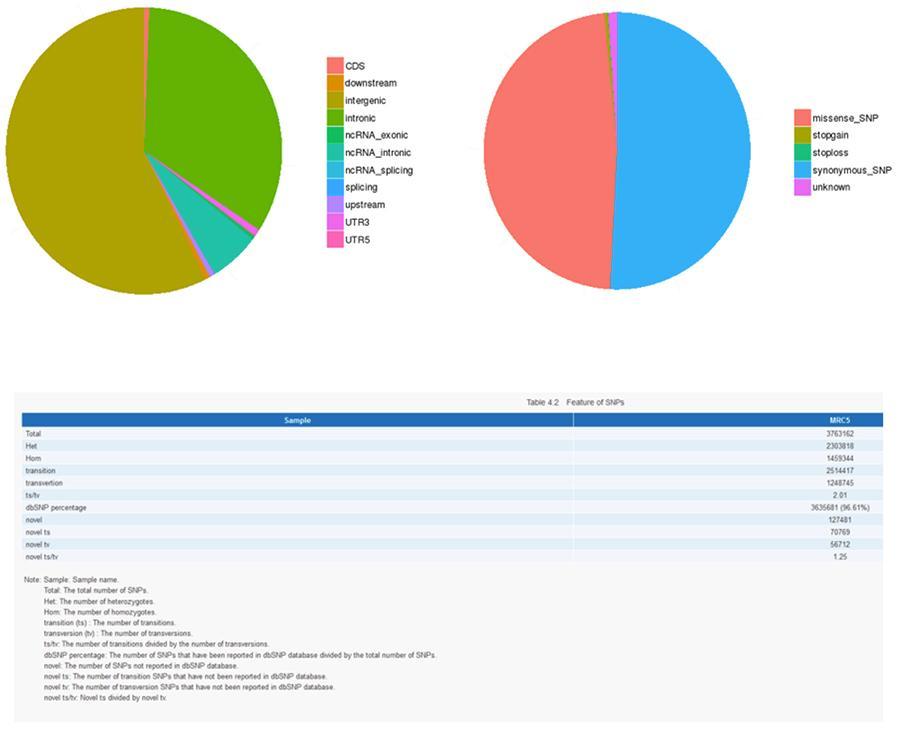
The results of the analysis of the various types of variants are shown below.
Single nucleotide variants (SNP) and short insertions / deletions (InDels)
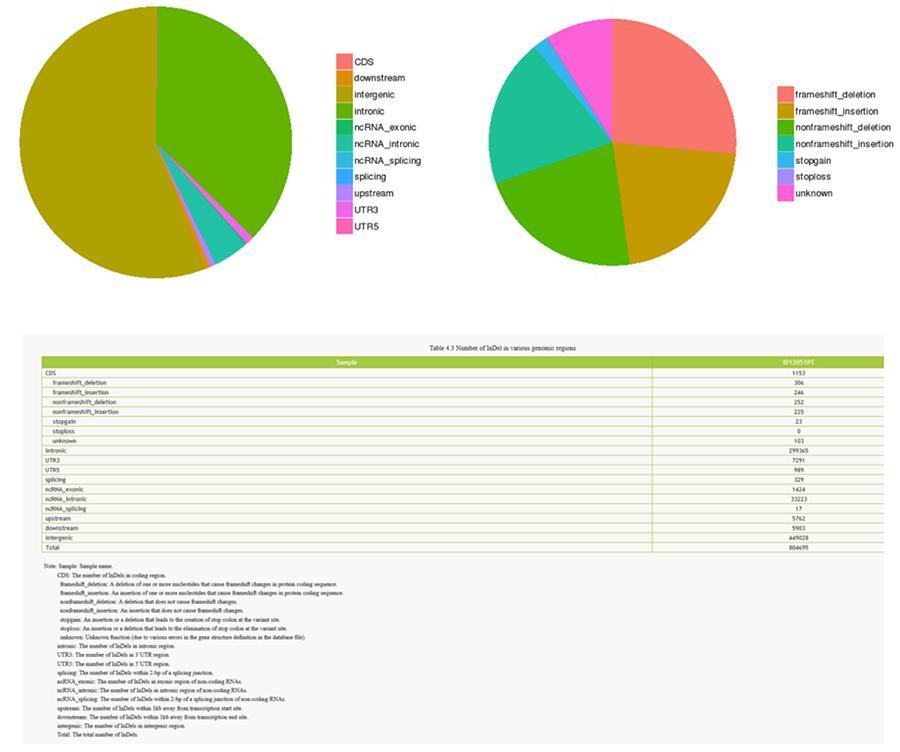
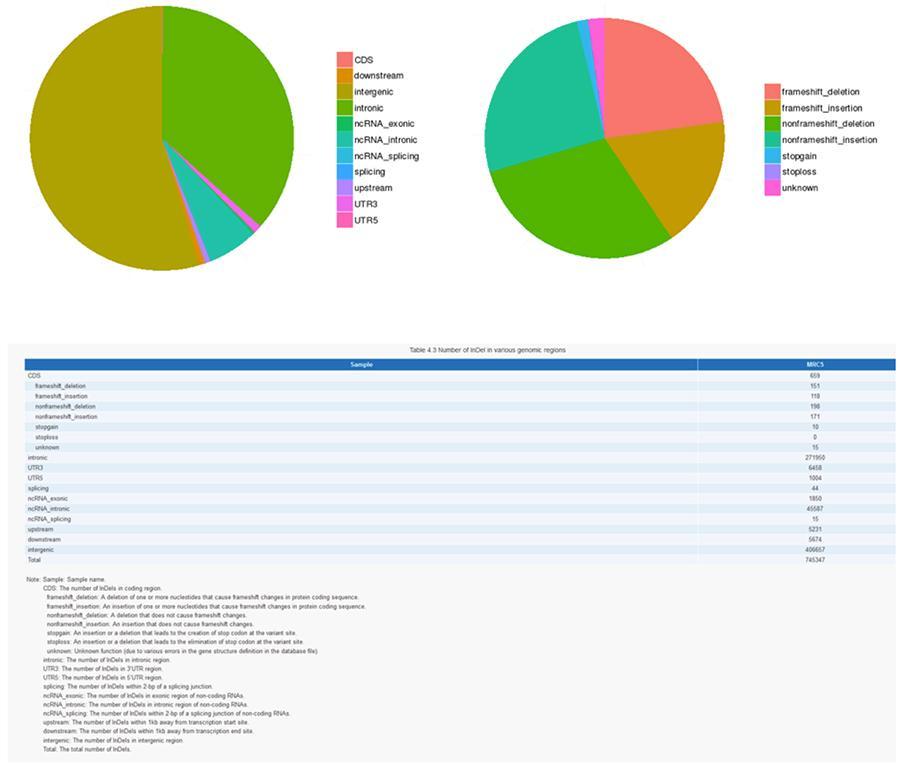
The variants of single DNA bases (SNP, pronounced 'snip') are polymorphisms, that is variations of the genetic material, borne by a single nucleotide. The 'InDels', on the other hand, are small insertions and deletions of less than 50bp in length and constitute another class of genomic variants in the human genome.
SNP (single nucleotide variants) - ID12051PT
SNP (single nucleotide variants) - MRC-5
INDELs (small insertions / deletions) - ID12051PT
INDELs (small insertions / deletions) - MRC-5
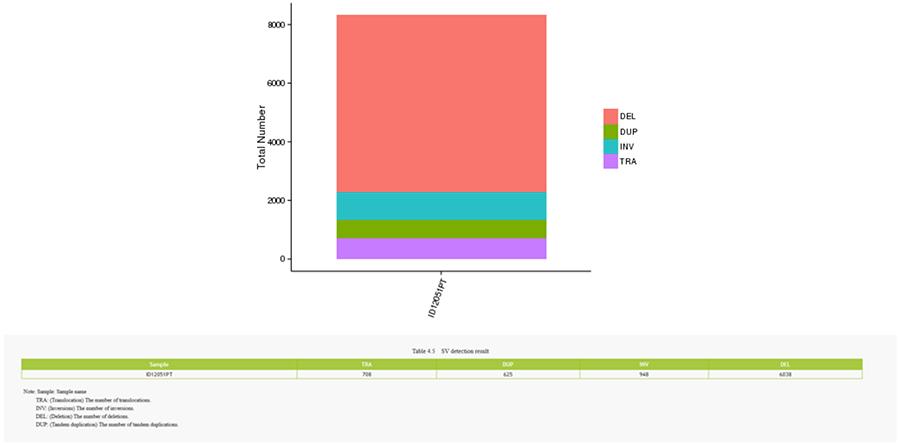
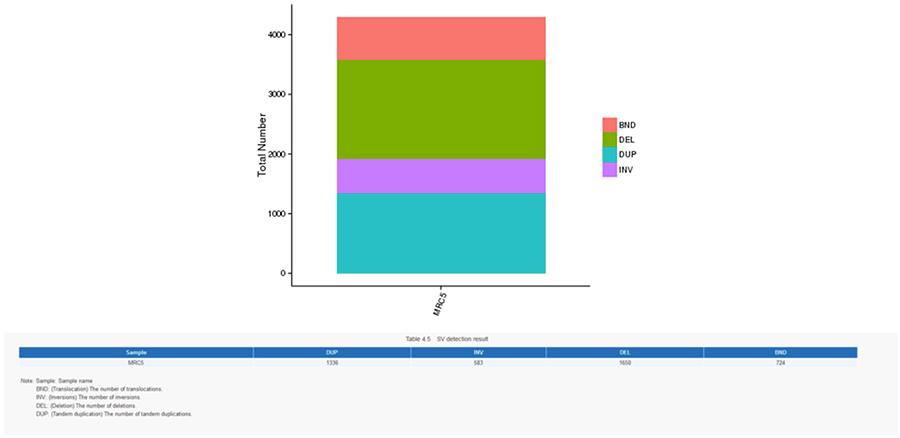
CNV (Copy Number Variants) and SV (Structural Variants)
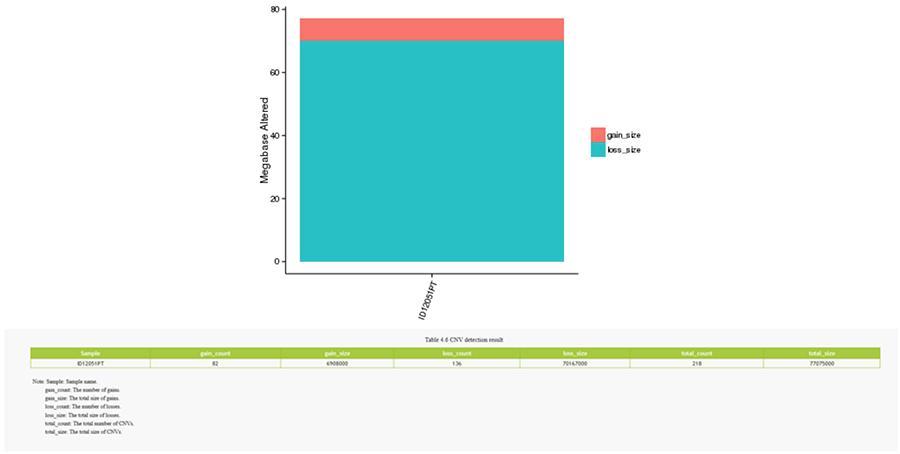
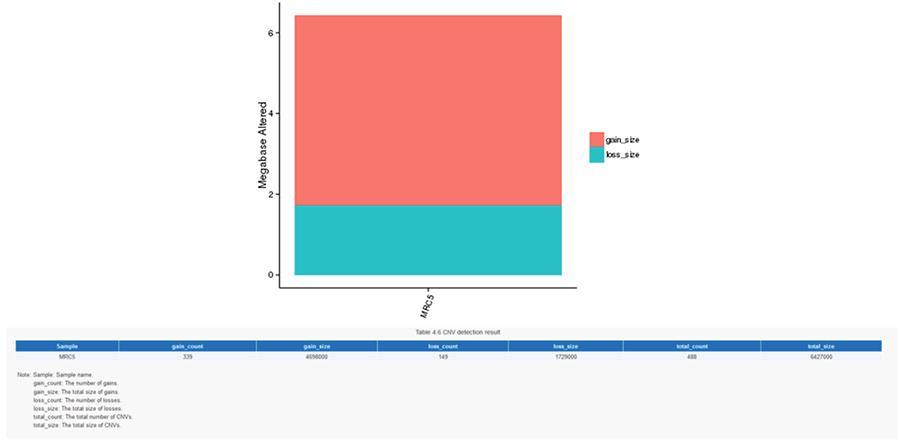
Variants in number of copies (CNV) are genomic variants due to variations in the number of copies of relatively large fragments (longer than 50 bp) between individual genomes. There are two types of CNV: 'gain' type (copy gain) and 'loss' type (loss of copies).
Structural variants (SV) are genomic variants with relatively large sizes (> 50 bp), including deletions, duplications, insertions, inversions and translocations.
CNV (variations in number of copies)
SV (structural variants)
Circular visualization of genomes (circos plot)
A graphical representation of the two genomes called 'circos plot' is shown below.
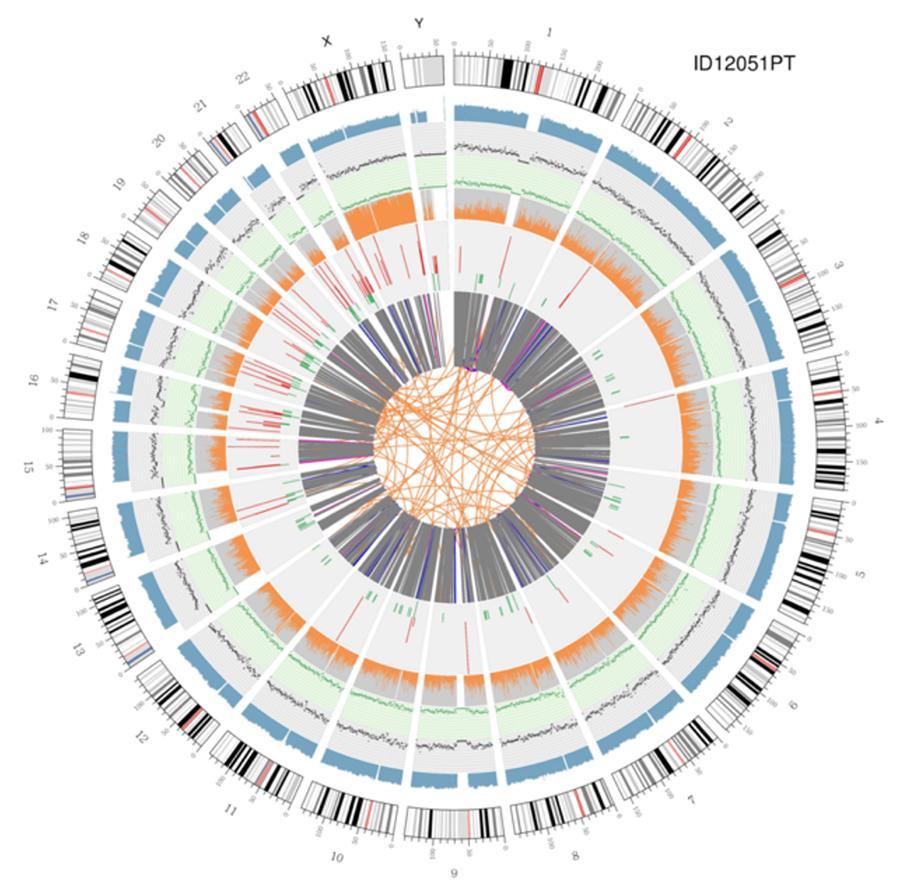
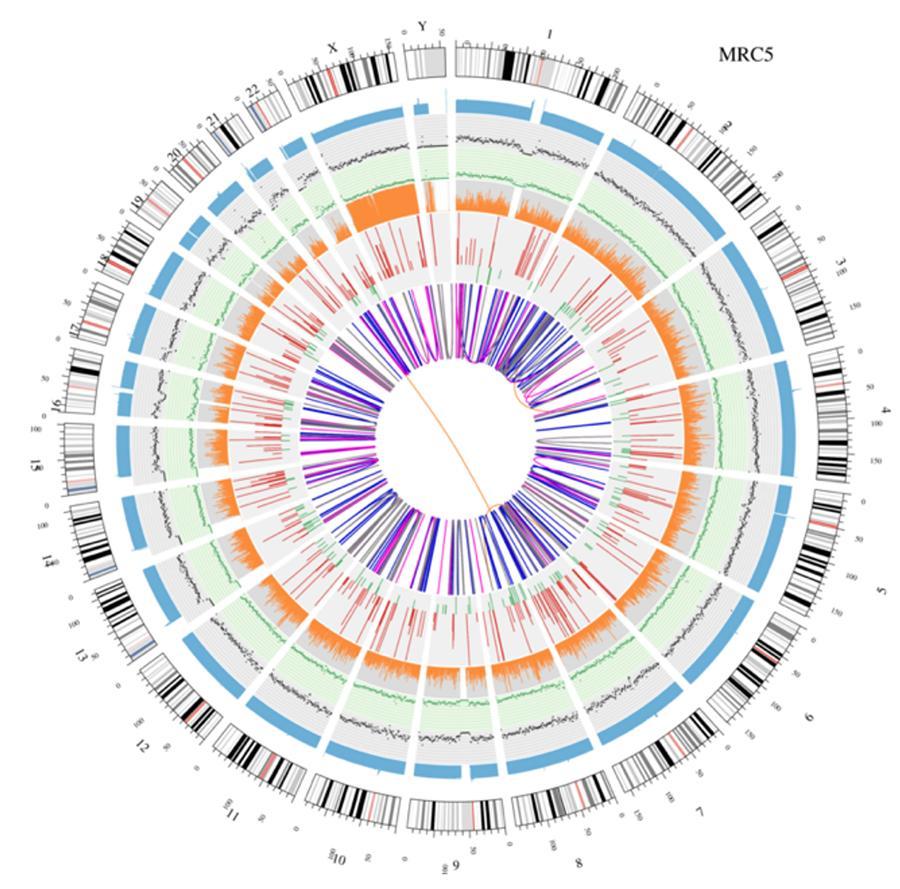
Meaning of the various concentric circles
- The outer circle (the first circle) is the chromosome number.
- The second ring (blue) represents the coverage of histogram-style reads. Each histogram is the average coverage of an area of 0,5 Mbp.
- The third ring (black) represents the density of the INDELs in a 'dispersion graph' style. Each black point is calculated as the number of INDELs (Small insertions / deletions) in a range of 1 Mbp.
- The fourth ring (green) represents the density snp in a 'dispersion graph' style. Each green dot is calculated as the number of SNPs (single nucleotide variants) in a range of 1 Mbp.
- The fifth ring represents the proportion of homozygosity (orange) and heterozygosity (gray) SNP in histogram style. Each histogram is calculated from a 1 Mbp region.
- The sixth ring represents the inference of CNV (Variants in number of copies). Red means gaining pieces of DNA and green means loss.
- The most central ring represents the inference of SV (Structural Variants) in the exonic and splicing regions. BETWEEN (orange, translocations), INS (green, insertions), DEL (deletions, gray), DUP (duplications, pink) and INV (inversions, blue).
Conclusions
The two genomes show important differences. In particular, the vaccine genome turns out to be heavily changed compared to that of the cell line deposited in 1966. It must be said that we did not have the possibility to sequence the cell line that is used by GlaxoSmithKline for the production of the Priorix Tetra vaccine, but that of the stock original. Pharmaceutical companies that use cell lines for the production of drugs and vaccines, have their MRC-5 line but this does not change our suspicions, that is the fact that the control of genetic stability is not required periodically and this is very serious in our opinion , above all because it is allowed to be present without a limit.

