First peer review publication on MPRV vaccines (Priorix Tetra)

We are finally there, after almost two years the first publication in peer review of our analyzes comes out and many others will follow.
Here we try to summarize in these first pages, in a very discursive and non-technical way: what has been published, what validity it has and why it is important for our investigation of vaccines.
(On the other hand, pages 3 to XNUMX are dedicated to a technical study, leaving the evaluation of the article itself to those working in the sector)
What published on "F1000 Research" 1 it is the result of the initial part of the work carried out on behalf of the Corvelva Association by one of the laboratories in charge of the analyzes. Recall - because more than two years have passed since the beginning of this work and many other results have been added to the initial ones - that the the first major issue that we found ourselves having to investigate was the abnormal amount of human DNA found in the vaccines analyzed.
In both the quadrivalent MPRV vaccines analyzed, quantities of 1 to 2,7 micrograms / vial were initially found (as per publication in question), and this led us to publicly and immediately report this result because, simply, it was not expected that a this amount of DNA was present in a vaccine.
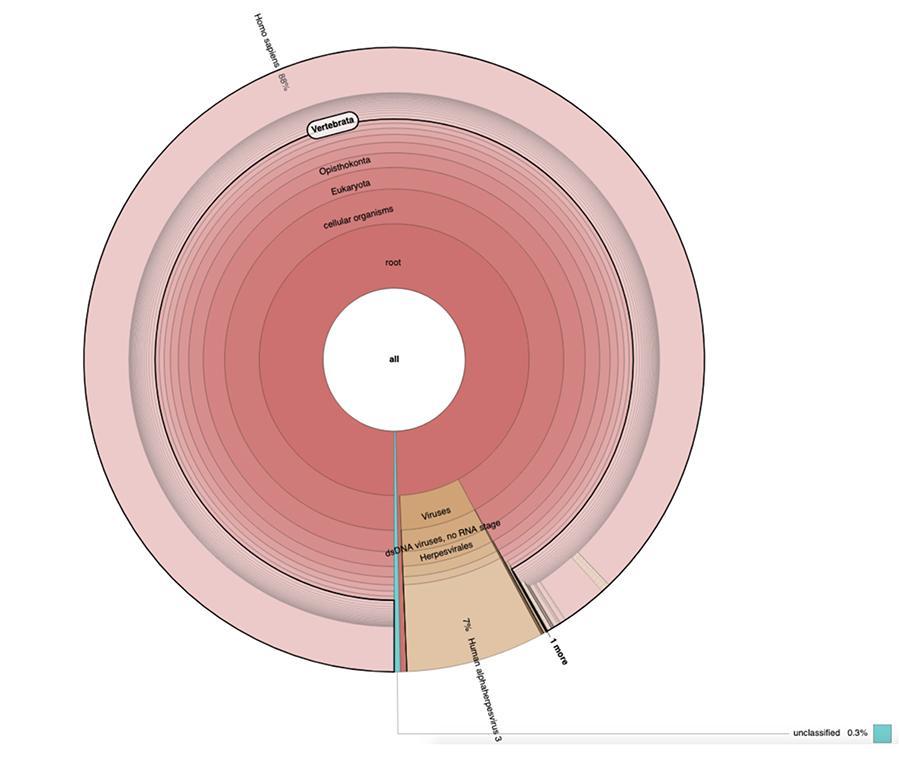
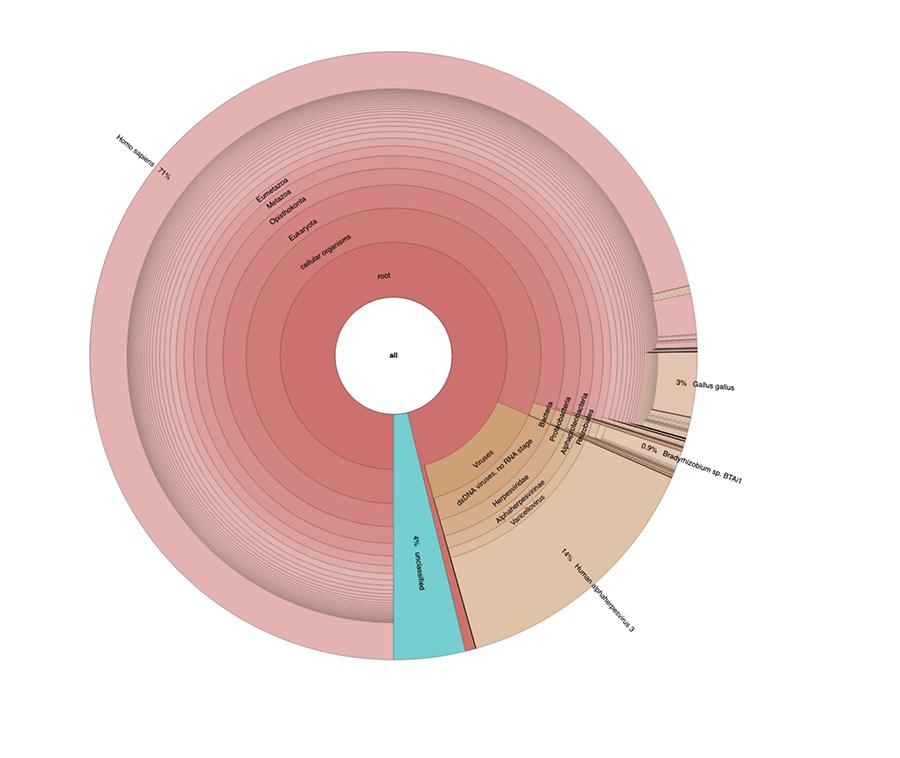
Apart from the considerations and conclusions to which the work reaches, which are strictly technical and therefore understandable only to those who do research in the field of metagenomics, what is observed in the graphs is that the two vaccine samples were found to contain a high percentage of readings of human DNA in addition to those expected from the genome of the chickenpox virus (Human alphaherpes virus 3), only detectable of the four, as a DNA-seq analysis was presented in the article.
However, we would like to emphasize that later the quantities of DNA found and confirmed with the same method that is now validated here were even higher: up to 3,7 micrograms per vial, leading to a significant difference between batch and batch.
In fact, in our report disclosed on 22.12.2018 2 results have been reported for further lots analyzed after those discussed in the article, then further confirmed by interlaboratory analyzes which are still being published.
Therefore, what interests us most about this publication is that it validates the method used, puts an important point on the discussions on the "type" of analyzes carried out, and consequently decisively confirms all the work done subsequently with the NGS method: the insights on the type of genetic material contained, the presence of adventitious viruses, the great absence of attenuated viruses that should instead be present and the quantity instead out of control (also because it is very different from sample to sample) of the human DNA present, the mutant population , phages, DNA from other species, and gradually all the results that you find summarized on our site. 3
Everything that, from the point of view of biological content, we have denounced in recent years, slavishly reporting the results to the control bodies, takes on a more relevant scientific connotation (even if, we reiterate it again, it was not the peer-reviews that had to worry but the data presented, very serious in their content and in their possible implications for human health). However, now that the publication of the method has been made, we will demand to obtain the answers that have not yet arrived.
These results conclusively confirm the presence of fetal DNA in Priorix tetra vaccines, in quantities varying between the various batches, indicating a poor quality control of these pharmaceutical products.
We also recall the report on the sequencing of the entire MRC-5 genome published on the Corvelva website on 27.09.2019 4 in which the profound modification of this DNA is evident also in genes associated with the development of tumor pathologies (other data being published).
The contaminating fetal DNA present in all the samples analyzed in variable quantities (therefore uncontrolled) is up to 300 times higher than the limit set by the EMA for carcinogenic DNA (10 ng / dose, corresponding to the DNA contained in about 1000 cancer cells, obtained on the basis of a statistical calculation, while the precautionary limit is 100 pg / dose) limit which must necessarily be applied also to the fetal DNA that inevitably contaminates the Priorix Tetra.
It follows that this vaccine must be considered defective and potentially dangerous for human health, in particular of the pediatric population much more vulnerable to genetic and autoimmune damage due to immaturity in the shelter systems.
As anticipated, the following is a more "technical" and difficult to understand for non-professionals, therefore we have decided, also for transparency, to attach to this document also the "EMA - NGS Dossier Discussion of the results obtained from the vaccine quality survey". We only had to extrapolate the disclosed part, more than 50 pages of files compared to 200 of the NGS, since much of the information contained and registered with regulatory bodies must remain confidential. The harsh law of science requires that data be published in a magazine only if unpublished and we, having several other publications in progress, do not want to put them at risk.
Finally, to avoid any misunderstanding, we want to mention, from the publication, the part of the "Declaration of financing":
“The metagenomic sequencing B1 and B2 was funded by Corvelva (non profit association, Veneto, Italy), as part of a service contract with the laboratory. No other contributions were involved in supporting the work. The funders had no role in the study design, in the collection and analysis of the data, in the decision to publish or in the preparation of the manuscript. "
attachments:
- Publication - Do you cov me? Effect of coverage reduction on metagenome shotgun sequencing studies
- CORVELVA-thread-NGS-EMA-ita
- PDF - First peer review publication on MPRV vaccines (Priorix Tetra)
In the article “Do you cov me? Effect of coverage reduction on metagenome shotgun sequencing studies "
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7059852/
The authors tackle the technical-methodological question of whether it is possible to use a massive parallel metagenomics approach with low reading coverage to characterize complex biological matrices. Estimates of diversity, species abundance and the ability to reconstruct the de novo metagenome in terms of length and completeness are calculated, in order to understand how much the decrease in sequencing depth, varied by randomly sub-sampling the sequencing reads, can The results show that the diversity indices of prokaryotic, eukaryotic and viral complex communities can be accurately estimated with 500.000 reads or less, although particularly complex samples may require 1.000.000 reads. On the contrary, a project involving the reconstruction of the metagenome and the genes it contains requires more than 1.000.000 reads.
Among the various and very different complex matrices subjected to massive metagenomic analysis, two biological medicines were included, that is two different lots of live attenuated MPRV vaccine used for immunization against measles, mumps, rubella and chicken pox in children. DNA was extracted from the vaccines, genomic libraries were then built using standard commercial protocols and massive sequencing with Illumina technology was carried out.
Apart from the considerations and conclusions to which the work reaches, which are strictly technical and therefore understandable only to those who do research in the field of metagenomics, what is observed in the pie charts contained in the 'Extended data' (https://osf.io/wq395/ samples B1 and B2) is that the two vaccine samples were found to contain a high percentage of human DNA readings in addition to those expected of the genome of the varicella virus (Human alphaherpes virus 3), the only detectable among the four, being in the article a DNA-seq analysis was presented.
71% of the readings in one batch and 88% in the other are of human origin, presumably deriving from the cell line of fetal origin MRC-5 (remember that subsequent analyzes have confirmed that the line is MRC5) in which live attenuated rubella and chicken pox viruses are grown during vaccine preparation. Furthermore, as happened in the different lots of the same MPRV vaccine tested by Corvelva between 2017 and 2019, the quantity of extracted DNA is of the order of the microgram.
In the vaccination lots tested with the same protocols and technology reported in the materials and methods of the article, the quantities detected ranged between 1 and almost 3 micrograms per vial, quantities varying between one batch and another, but always significant
In the report disclosed by Corvelva on 22.12.2018 the following results were reported for further lots analyzed after those discussed in the article, then further confirmed by interlaboratory analysis still being published:
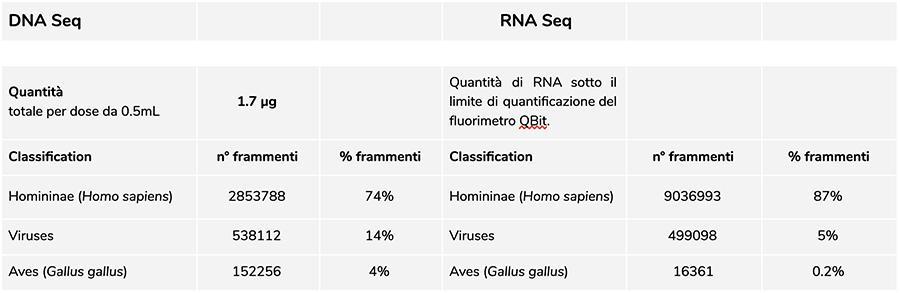
Priorix Tetra lot. A71CB205A (June 2018) - DNA analysis
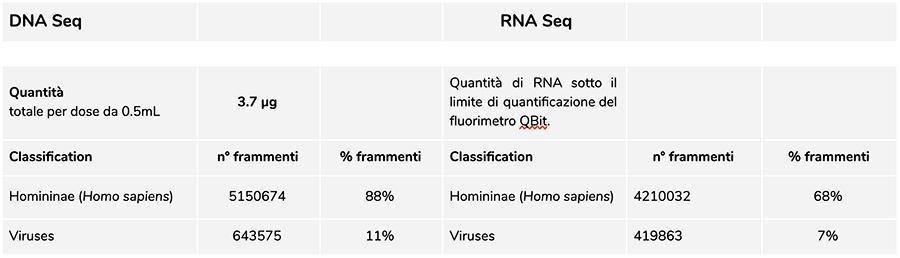
Priorix Tetra lot. A71CB256A (December 2018) - DNA analysis
DNA analysis
The measurement of the concentration of DNA with QuBit fluorimeter showed that lot A71CB205A contains a quantity of 1.7 µg gDNA totals per 0.5mL dose, calculated as follows: 9.41ng / µl (concentration determined at QuBit) x 45 (final resuspension volume of DNA after extraction, expressed in microliters) x 4 (the starting volume subjected to the extraction procedure is ¼ the volume of the dose contained in the entire vial equal to 0.5mL).
The measurement of the concentration of DNA with QuBit fluorimeter showed that lot A71CB256A contains a quantity of gDNA of 3.7 µg total per 0.5mL dose, calculated as follows: 40.8 ng / µl (concentration determined at QuBit) x 55 (final resuspension volume of DNA after extraction expressed in microliters) x 5/3 (the starting volume subjected to the extraction procedure is 300 µl out of 500 µl of suspension).
The human DNA found in this lot has a relative ratio of about 8 to 1 compared to the chickenpox DNA (see following results of the classification of the DNA-seq fragments, in which it emerges that 88% of the total of the sequenced DNA fragments is of human origin, and 11% are of the varicella virus genome). Considering that the NGS is a quantitative technology, the fluorimetric quantification of the total DNA extracted from the vaccine (e.g. lot. A71CB256A = 3,7 micrograms per dose), associated with the consideration of relative quantification made above (8: 1), allows us to say that human DNA could be around 2,9 micrograms per dose, compared to about 740 nanograms of chickenpox DNA. It is also plausible that at least a portion of the high molecular weight DNA that is seen on gel may be high molecular weight human DNA.
RNA analysis
The amount of RNA contained in the vial of vaccine lot A71CB256A was found to be approximately 200ng.
The RIN equal to 8 indicates an excellent quality RNA and an intact eukaryotic RNA, since both the 18S and 28S peaks typical of eukaryotic RNA are present.
The answers to our questions forwarded to the regulatory agencies over time are of great importance. Currently, the agencies have not yet answered the questions regarding the results of the full analyzes delivered to the EMA and AIFA.
Excerpt from the ema answer to our question regarding the safety of mrc-5 residues in the priorix tetra vaccine (EMA request reference ask-43967 3 august 2018) - “Based on the published information, Priorix Tetra contains viral strains produced separately in chicken embryo cells (mumps and measles) or in human diploid cells MRC-5 (rubella and chicken pox). The cell lines used for Priorix Tetra include human diploid cell lines that cannot continuously divide. Note that, according to the European Pharmacopoeia, MRC-5 diploid cell lines are not tumorigenic, as demonstrated by decades of use and control, and therefore a maximum limit for the DNA of MRC-5 cells does not apply "
To date, no evidence has been provided (neither in terms of certificates of analysis on the quality of the product, nor of scientific reference literature for the EMA) of these controls which guarantee that it is appropriate not to apply a maximum limit.
In the FDA guideline "Guidance for Industry: Characterization and Qualification of Cell Substrates and Other Biological Materials Used in the Production of Viral Vaccines for Infectious Disease Indications" 6 it is reported that:
- a strain of diploid cells should always remain diploid. If these characteristics are not stable, it is necessary to demonstrate that instability does not adversely affect the production or conformity of the product.
- for widely used human diploid cell strains, such as MRC-5 and WI-38 cells, measurement of residual DNA may not be necessary because we do not consider the residual DNA of these human diploid cells to be a safety concern
- residual DNA should be limited for non-tumorigenic continuous cells, such as low pass VERO cells, less than 10 ng / dose for parenteral inoculation as recommended by WHO
And in the WHO guideline “Annex 3 - Recommendations for the evaluation of animal cell cultures as substrates for the manufacture of biological medicines and for the characterization of cell banks. Replacement of Annex 1 of the WHO series of technical reports, n. 878 " 7 we add: (...) considerable experience has been accumulated on the cytogenetics of WI-38 and MRC-5 since the 60s
and to support this experience, the following articles are listed:
- Jacobs JP. Updated results on the karyology of the WI-38, MRC-5 and MRC-9 cell strains. Developments in Biological Standardization, 1976, 37: 155–156.
- Jacobs JP. et al. Guidelines for the acceptability, management and testing of serially propagated human diploid cells for the production of live virus vaccines for use in man. Journal of Biological Standardization, 1981, 9: 331–342.
- Petricciani JC et al. Karyology standards for rhesus diploid cell line DBS-FRhL-2. Journal of Biological Standardization, 1976, 4: 43–49.
- Schollmayer et et al. High resolution analysis and differential condensation in RBA-banded human chromosomes. Human Genetics, 1981, 59: 187–193.
- Rønne M. Chromosome preparation and high resolution banding techniques: a review. Journal of Dairy Science, 1989, 72: 1363–1377.
It can be clearly observed that the reference literature, to maintain that diploid cells used for the production of vaccines are safe from the point of view of genetic stability, it is obsolete. Already 40 years ago the first genetic anomalies were found, considered negligible for the safety of vaccines, and from what reported in the WHO guideline, since then no updates have been made with the new sequencing technologies, in particular in NGS, which is moreover economic and rapid, with the consequence that in the vaccines administered for decades, the presence of increasingly progressively genetically modified DNA in uncontrolled quantities has been allowed by agencies. In this regard, see the report on the sequencing of the entire MRC-5 genome published on the Corvelva website on 27.09.2019. 8 in which the profound modification of this DNA is evident also in genes associated with the development of tumor pathologies. (data being published)
An extract from the letter by Dr. T. Deisher, world expert in the use of stem cells for therapeutic purposes and gene therapy, is reported, which highlights the concern of the risks associated with the use of vaccines contaminated with residues of human fetal cells:
Dr. T. DEISHER (letter to the rulers - April 8, 2019) 9 - (...) injecting our children with human fetal DNA contamination carries the risk of causing two well-established diseases:
- insertional mutagenesis: human fetal DNA is incorporated into the baby's DNA causing mutations. Gene therapy using homologous small fragment recombination has shown that quantities as small as 1,9 ng / mL of DNA fragments result in the insertion of stem cells into the genome in 100% of injected mice. The levels of human fetal DNA fragments in our children after vaccination with MMR, VARIVAX (chicken pox) or hepatitis A vaccines reach levels above 1,9 ng / ml.
- autoimmune disease: fetal human DNA stimulates the immune system's reaction to attack the body of the baby.
Our results greatly strengthen the experimental observations of Dr. Deisher and above all the fact that the contaminating fetal DNA present in all the samples analyzed in variable (therefore uncontrolled) quantities is up to 300 times the EMA limit for carcinogenic DNA (10 ng / dose, corresponding to the DNA contained in about 1000 cancer cells, obtained on the basis of a statistical calculation, while the precautionary limit is 100 pg / dose) limit that must necessarily be applied also to fetal DNA that inevitably contaminates the Priorix Tetra.
It follows that this vaccine must be considered defective and potentially dangerous for human health, in particular of the pediatric population much more vulnerable to genetic and autoimmune damage due to immaturity in the shelter systems.
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7059852/
- https://www.corvelva.it/speciale-corvelva/vaccinegate/analisi-metagenomiche-su-priorix-tetra.html
- https://www.corvelva.it/speciale-corvelva/vaccinegate.html
https://www.corvelva.it/speciale-corvelva/vaccinegate-en.html - https://www.corvelva.it/speciale-corvelva/vaccinegate/sequenziamento-del-genoma-completo-di-mrc-5-contenuto-in-priorix-tetra.html
- https://www.corvelva.it/speciale-corvelva/vaccinegate/analisi-metagenomiche-su-priorix-tetra.html
- https://www.federalregister.gov/documents/2010/03/04/2010-4553/guidance-for-industry-characterization-and-qualification-of-cell-substrates-and-other-biological
- https://www.who.int/biologicals/vaccines/TRS_978_Annex_3.pdf
- https://www.corvelva.it/speciale-corvelva/vaccinegate/sequenziamento-del-genoma-completo-di-mrc-5-contenuto-in-priorix-tetra.html
- https://www.corvelva.it/approfondimenti/notizie/mondo/lettera-aperta-ai-legislatori-sul-dna-fetale-nei-vaccini-theresa-deisher.html

